 
So overall, zinc finds it easier to lose its valence electrons than copper, and so is more reactive, and thus can displace copper. Zinc has more protons than copper but the other two conditions outweigh this one. Zinc is more reactive than copper because its valence electrons are farther away from the nucleus and it has more shells. there are fewer protons in the nucleus (less charge) to pull the electrons towards them.These shells shield the valence electrons from the charge of the nucleus (protons). there are more electron shells between the nucleus and the valence electrons.So it is easier to ‘let go’ of the electrons. When they are farther away, the attraction of these electrons (negative charge) to the protons (positive charge)in the nucleus will be reduced. the valence electrons are further away from the nucleus.It is easier to lose valence electrons and thus be more reactive, if: The more reactive a metal is, the more easily it loses its valence electrons. We can see that the more reactive metal, zinc, has lost electrons and the less reactive metal, copper, has gained electrons. Zn(s) – 2e-–> Zn2+(aq) the zinc oxidised (lost electrons)Ĭu2+(aq) + 2e-–> Cu(s) the copper reduced (gained electrons) Here, the zinc, being the more reactive metal, has clearly displaced copper to form zinc sulfate Zinc + Copper(II)sulfate -–> zinc sulfate + copper Let’s take another example, with an aqueous solution, and see how the metal ions form: This happened because aluminium is higher in the reactivity series than iron i.e. In the above, reaction the aluminium displaced the iron (think of it as ‘the aluminium stole the oxide and the iron was left alone’). Iron(III) oxide + aluminium -> iron + aluminium oxide Metals can also ‘compete’ in a similar way. 
We call this type of a reaction a displacement reaction. We can see this is a competition to see which halogen combines best with the metal. Previosuly, we saw that a more reactive halogen will replace a less reactive halogen in a metal halide. the aqueous ions of other listed metals. 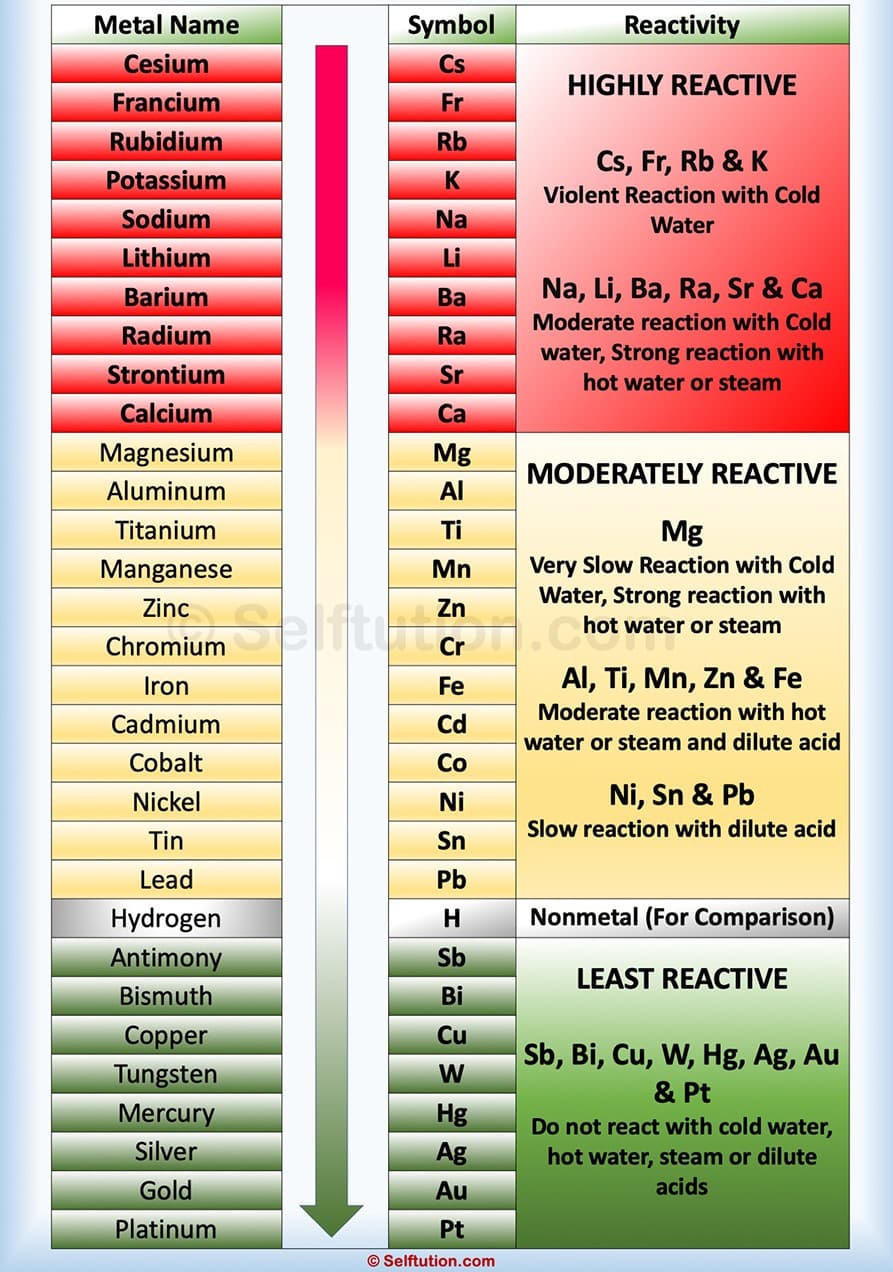
Compare the reactivity series to the tendency of a metal to form its positive ion, illustrated by its reaction, if any, with:.This is the reactivity series, from most reactive to least reactive (and a mnemonic to remember it!) : Only metals above hydrogen will react with water/steam and hydrochloric acid. We can see that the the above elements are put in their order of reactivity- the more reactive on the top (potassium, sodium, calcium etc) and the least reactive on the bottom (iron, hydrogen, copper). 
Reaction of metals and hydrogen with dilute (hydrochloric) acid: Reaction of metals and hydrogen with water or steam: We can put metals in order of their reactivity by investigating how well they react with water or hydrochloric acid.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
